 In my work as an arranging teacher at the conservatory, I often encounter students claiming that a specific way of notating chords is wrong. Apparently they have learned to write chords in a certain way and they think that is the only correct way. That is a shame, they should have been told that there are a lot of differences in the way people use chord symbols.
In my work as an arranging teacher at the conservatory, I often encounter students claiming that a specific way of notating chords is wrong. Apparently they have learned to write chords in a certain way and they think that is the only correct way. That is a shame, they should have been told that there are a lot of differences in the way people use chord symbols.
A nice way to illustrate this, is to look at some famous books with standards. Let’s compare the Real Book and the New Real Book, two of the most well-known collections of standards, used by millions and millions of musicians. Here is the first part of Misty, according to the Real Book:
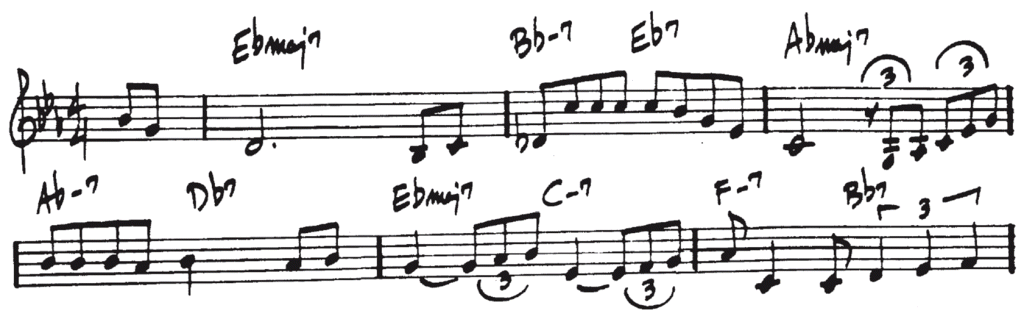
And here is the version of the New Real Book:

As you can see, the actual chords in the books differ. In the New Real Book the chords are slightly more complicated. But that is not our concern here. We are interested in the conventions used in notation chords. You may be surprised to see the many differences between these two versions. The books use different ways to notate a minor chord (F_7
versus FMI7
) and a major seventh (E@maj7
versus E@ma7
). Furthermore, the books deal with capitalization and superscript differently. The Real Book uses upperscript only for extensions, but the New Real Book places the @
for the root of the chord high as well. Thus, the first one writes B@7
and the latter B&7
. Surprisingly, the New Real Book uses capitals for all extensions (something you hardly ever see in other publications).
If there are so many obvious differences between these two popular books, standardization of chord symbols is still a long road ahead of us.
